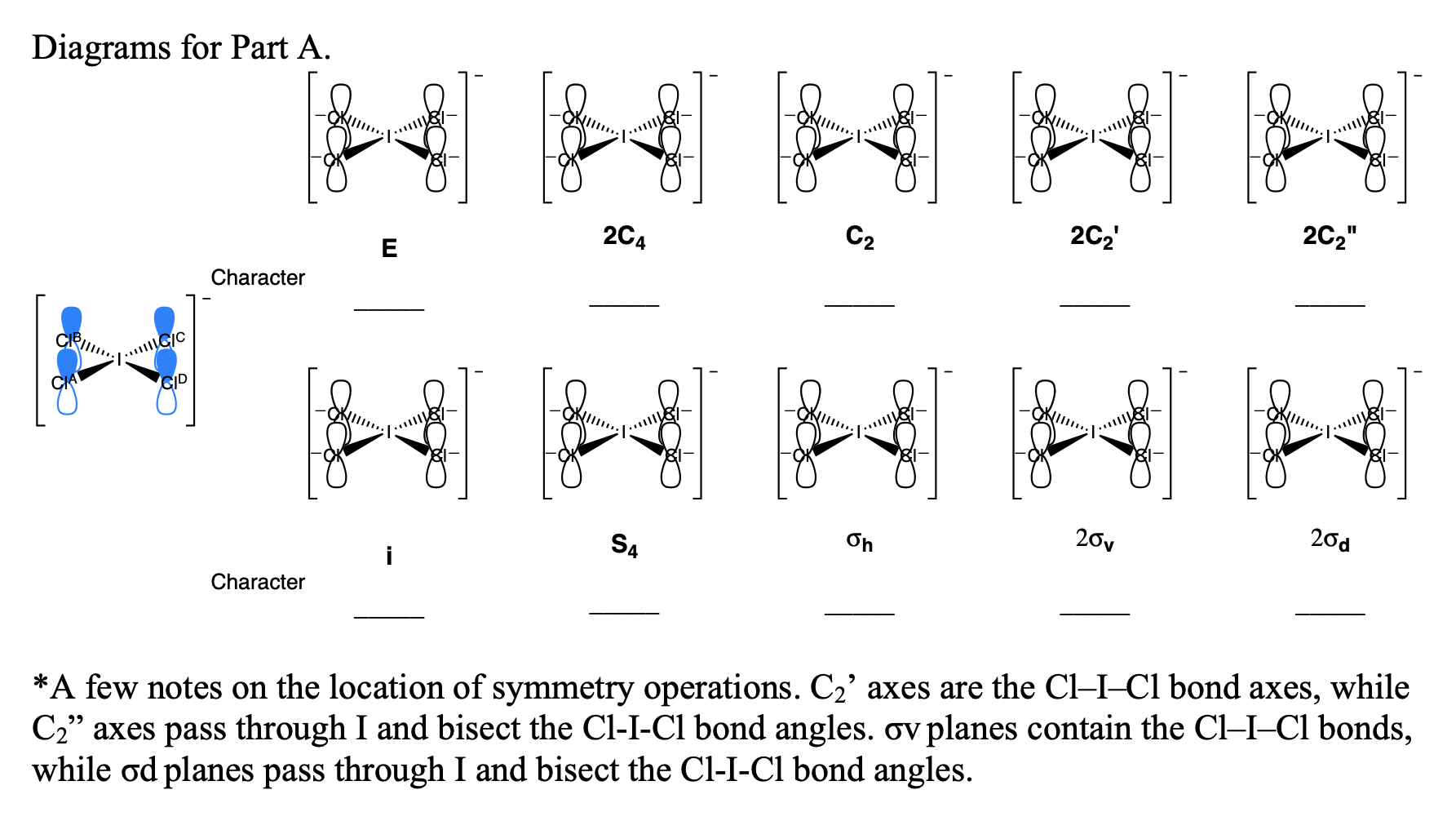
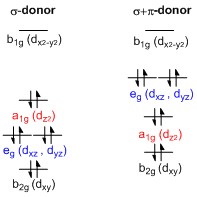
Square Planar Complexes (5.2.7) | Edexcel International A Level Chemistry Revision Notes 2017 | Save My Exams

Square planar versus square pyramidal copper(II) complexes containing N3O moiety: Synthesis, structural characterization, kinetic and catalytic mimicking activity - ScienceDirect

Three examples of square planar da complexes. C204 is the oxalate g r o... | Download Scientific Diagram

Seesaw Molecular Geometry - Seesaw Shaped Molecules, Lone pairs, Examples & Hybridisation of Seesaw Molecular Geometry

Square Planar Complexes (5.2.7) | Edexcel International A Level Chemistry Revision Notes 2017 | Save My Exams
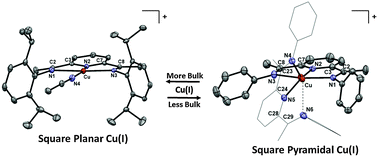
Square planar Cu(i) stabilized by a pyridinediimine ligand - Chemical Communications (RSC Publishing)
![16-Square planar [4]-fold geometry and its off-plan distortion along... | Download Scientific Diagram 16-Square planar [4]-fold geometry and its off-plan distortion along... | Download Scientific Diagram](https://www.researchgate.net/publication/313641543/figure/fig17/AS:669448675545111@1536620410383/Square-planar-4-fold-geometry-and-its-off-plan-distortion-along-the-C-4-axis.png)
16-Square planar [4]-fold geometry and its off-plan distortion along... | Download Scientific Diagram